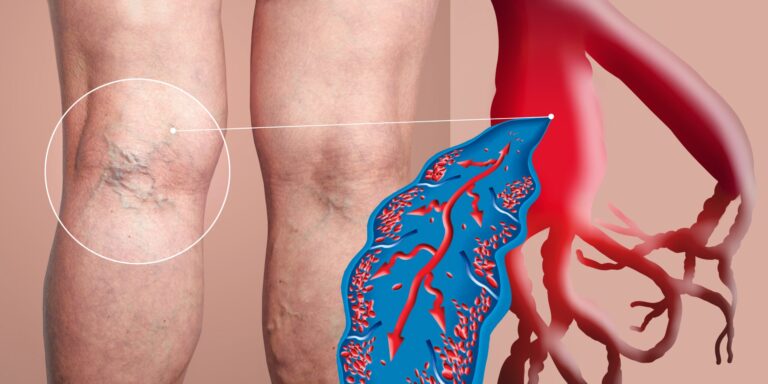
Deep vein thrombosis (DVT) is a serious condition that can lead to life-threatening complications. Understanding its symptoms and risk factors is crucial for prevention and early intervention. This guide will explore what DVT is when it becomes an emergency, its symptoms, who is at risk, and how to prevent it.
What is DVT?
Deep vein thrombosis occurs when a blood clot forms in the deep veins, most commonly in the legs. This condition can obstruct blood flow, leading to swelling and pain. The clot may also dislodge and travel to the lungs, resulting in a pulmonary embolism (PE), which can be fatal. DVT is part of a broader category known as venous thromboembolism (VTE) and represents a significant health risk, particularly among certain populations.
When Deep Vein Thrombosis is an Emergency
Recognizing when DVT becomes an emergency is vital. If you experience any of the following symptoms, seek immediate medical attention:
- Severe leg swelling: Particularly if one leg is significantly larger than the other.
- Intense pain or tenderness: Often described as a throbbing sensation in the affected leg.
- Skin changes: Look for discoloration or warmth in the leg.

Signs of pulmonary embolism: These include sudden shortness of breath, chest pain, rapid heart rate, or coughing up blood.
Deep Vein Thrombosis Symptoms
DVT can manifest through various symptoms, though some individuals may remain asymptomatic. Common signs include:
- Swelling: Typically in one leg, but it can occur in both.
- Pain or tenderness: Often localized to the calf or thigh.
- Skin discoloration: Affected areas may appear red or blue.
- Warmth: The skin over the affected area may feel warmer than the surrounding skin.
It is important to note that about half of all DVT cases present no symptoms at all, making awareness crucial for those at risk.
Who’s at Risk for Deep Vein Thrombosis?
Several factors increase the likelihood of developing DVT:
- Prolonged inactivity: Long periods of sitting or lying down can slow blood flow.
- Surgery or injury: Procedures involving the legs or pelvis increase risk due to potential vein damage.
- Hormonal factors: High estrogen levels from pregnancy, birth control pills, or hormone replacement therapy can elevate risk.
- Obesity: Excess weight places additional pressure on veins.
Age and family history: Individuals over 60 or with a family history of blood clots are more susceptible.
How to Prevent Deep Vein Thrombosis
Preventing DVT involves lifestyle modifications and awareness of risk factors:
- Stay active: Regular physical activity promotes healthy blood circulation. If you have a sedentary job, take frequent breaks to stand and stretch.
- Hydration: Drink plenty of fluids to keep blood flowing smoothly.
- Weight management: Maintaining a healthy weight reduces pressure on your veins.
- Avoid tobacco: Smoking cessation is crucial as tobacco use contributes to vascular problems.
Be cautious with medications: Discuss with your doctor any hormonal treatments you may be taking and their potential risks concerning DVT.
By understanding DVT’s symptoms and risk factors, individuals can take proactive steps to protect themselves from this serious condition. Regular check-ups with healthcare providers are also essential for those at higher risk.
Also read: Nerve damage from sitting too much can be prevented, here’s HOW!