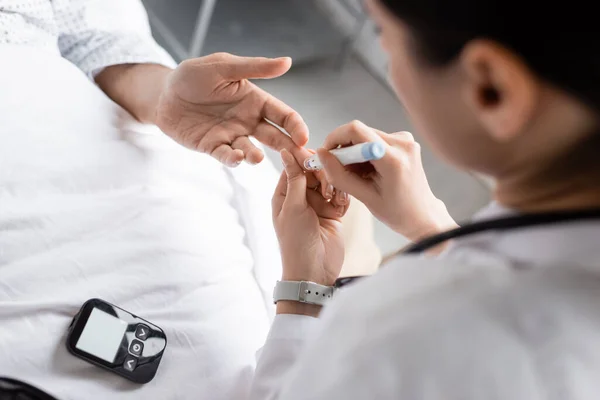
Long-term high blood sugar ( hyperglycemia ) has been linked to an Increased Risk of Hand and Wrist Problems (upper-limb musculoskeletal conditions) in Diabetics. A study published in the International Journal of Epidemiology demonstrated the association between long-term hyperglycemia and adiposity in Europeans, highlighting hyperglycemia as a significant factor in the incidence of conditions such as Dupuytren’s disease, frozen shoulder, trigger finger, and carpal tunnel syndrome.
Another study consistently demonstrated a causal role of long-term high glycaemia in the etiology of upper limb musculoskeletal conditions.
Evidence suggests that hyperglycemia may accelerate non-enzymatic glycosylation and abnormal collagen deposition in per-articular connective tissue, leading to conditions such as adhesive capsulitis of the shoulder, carpal tunnel syndrome, Dupuytren’s contracture, trigger finger, and limited joint mobility, which are more commonly occurring in patients with diabetes.
Therefore, it is important for clinicians treating diabetic individuals to consider the potential musculoskeletal complications associated with long-term hyperglycemia.
Key findings:
- High blood sugar levels directly increase the risk of all four conditions.
People with genetic mutations in a specific gene linked to blood sugar control are even more likely to develop these problems. - Body mass index (BMI) plays a complex role, increasing the risk of carpal tunnel syndrome but potentially protecting against Dupuytren’s disease.
- Smoking and alcohol consumption were also linked to some of the conditions.
This research highlights the importance of good blood sugar control in people with diabetes to prevent hand and wrist problems. Clinicians should be aware of the increased risk in these patients and monitor them closely for signs of these conditions.
How does hyperglycemia affect the musculoskeletal system
Hyperglycemia can affect the musculoskeletal system in various ways. Long-term hyperglycemia has been found to be a causal risk factor for various upper-limb musculoskeletal conditions such as Dupuytren’s disease, frozen shoulder, trigger finger, and carpal tunnel syndrome.
Evidence suggests that hyperglycemia may accelerate non-enzymatic glycosylation and abnormal collagen deposition in per-articular connective tissue, leading to conditions such as adhesive capsulitis of the shoulder, carpal tunnel syndrome, Dupuytren’s contracture, trigger finger, and limited joint mobility, which are more commonly occurring in patients with diabetes.
Additionally, diabetic patients may suffer from a wide range of musculoskeletal disorders that can cause pain and limit the exercise.
The bone quality is deteriorated in both types of diabetes along with hyperglycemia causing increased glycosylation of structural proteins.
Therefore, clinicians treating diabetic individuals should be aware of these complications in clinic, specifically those managing the care of GCK mutation carriers.
High Blood Sugar Linked to Increased Risk of Hand and Wrist Problems in Diabetics | Study source ALSO READ: Stevia Doesn’t Shake Up Gut Bugs: Study Finds Minor Microbiome Impacts After 12 Weeks