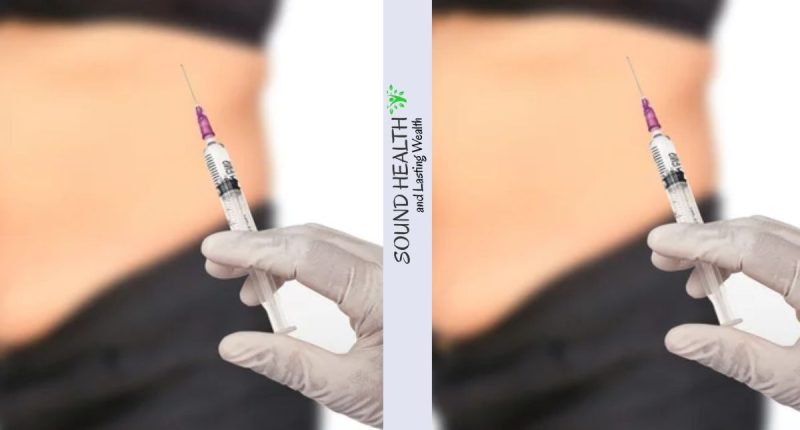
Fake Ozempic and Saxenda Weight Loss Jabs Found in UK, Causing Serious Side Effects – The Medicines and Healthcare products Regulatory Agency (MHRA) has issued a stark warning regarding the circulation of counterfeit Ozempic and Saxenda weight loss injections in the UK. These unauthorized products have been linked to a series of severe adverse reactions, including hypoglycemic shock, a potentially life-threatening condition characterized by dangerously low blood sugar levels.
Authorities have identified at least 16 individuals who have experienced adverse reactions after using the fake pens, with up to five requiring hospitalization for life-threatening complications. Investigations have revealed that these counterfeit products contain insulin, a substance that, when administered in inappropriate dosages, can lead to a precipitous drop in blood sugar levels, triggering a range of debilitating symptoms, including hypoglycemic shock.
The MHRA’s concerns stem from the increasing popularity of Ozempic and Saxenda, both of which are prescription medications approved for the treatment of type 2 diabetes. However, their effectiveness in promoting weight loss has led to a surge in demand, creating an opportunity for illicit manufacturers to produce and distribute counterfeit versions.
To combat this growing threat, the MHRA is urging the public to exercise extreme caution when purchasing or using Ozempic and Saxenda. These medications should only be obtained through legitimate healthcare providers, such as registered pharmacies. Consumers should never purchase these products from unauthorized sources, such as online marketplaces or social media platforms.
The MHRA also emphasizes the importance of seeking immediate medical attention if any adverse effects are experienced after using Ozempic or Saxenda. Healthcare professionals are urged to quarantine any suspected fake products and retain them for testing.
The emergence of counterfeit Ozempic and Saxenda highlights the growing challenge of safeguarding the pharmaceutical supply chain. As the demand for these medications continues to rise, it is crucial for regulatory agencies and healthcare providers to remain vigilant in identifying and combating counterfeit products to protect public health.
Everything about weight loss injection – How does it work?
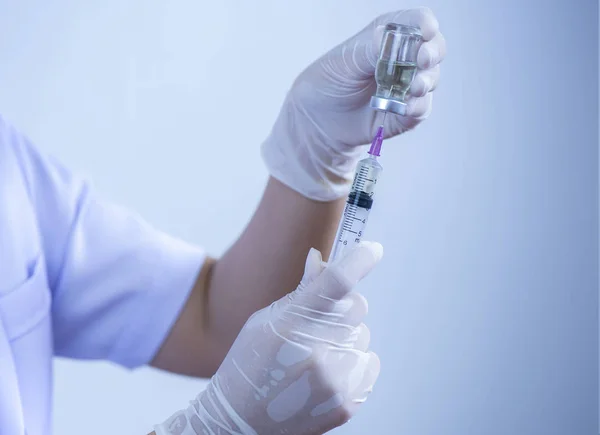
Weight loss injections have become increasingly popular in recent years as a way to help people lose weight and improve their health. These injections work by targeting hormones that affect your appetite and metabolism. The most common type of weight loss injection is a glucagon-like peptide-1 (GLP-1) receptor agonist. GLP-1 is a hormone that is naturally produced in the body and helps to regulate blood sugar levels and appetite. GLP-1 receptor agonists mimic the effects of GLP-1 and can help to reduce appetite and increase metabolism.
Here are some of the most common weight loss injections and how they work:
Liraglutide (Saxenda): Liraglutide is a GLP-1 receptor agonist that is injected once a day. It works by slowing down the emptying of the stomach and reducing appetite.
Semaglutide (Ozempic, Wegovy): Semaglutide is another GLP-1 receptor agonist that is injected once a week. It works in the same way as liraglutide, but it is also thought to increase insulin sensitivity, which can help to lower blood sugar levels.
Tirzepatide (Mounjaro): Tirzepatide is a dual agonist that targets both GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) receptors. GIP is another hormone that helps to regulate appetite and metabolism. Tirzepatide is injected once a week and has been shown to be more effective than liraglutide or semaglutide for weight loss.
Weight loss injections are not a magic bullet for weight loss. They should be used in conjunction with a healthy diet and exercise program. However, they can be a helpful tool for people who have struggled to lose weight on their own.
Here are some of the potential benefits of weight loss injections:
- Can help you lose weight and keep it off
- May improve blood sugar control in people with type 2 diabetes
- May lower blood pressure
- May improve cholesterol levels
However, there are also some potential risks associated with weight loss injections. These include:
- Nausea
- Vomiting
- Diarrhea
- Abdominal pain
- Constipation
- Low blood sugar
If you are considering using weight loss injections, it is important to talk to your doctor to weigh the risks and benefits. They can help you determine if weight loss injections are right for you and can monitor you for any side effects.
Resources:
MHRA warns of unsafe fake weight loss pens: GOV.UK
Continue to check our website soundhealthandlastingwealth.com for more articles of this kind. And, please use our comment section as well, we would love to hear from you.